Winners!
Public Vote: The Galaxy of the Little Brain
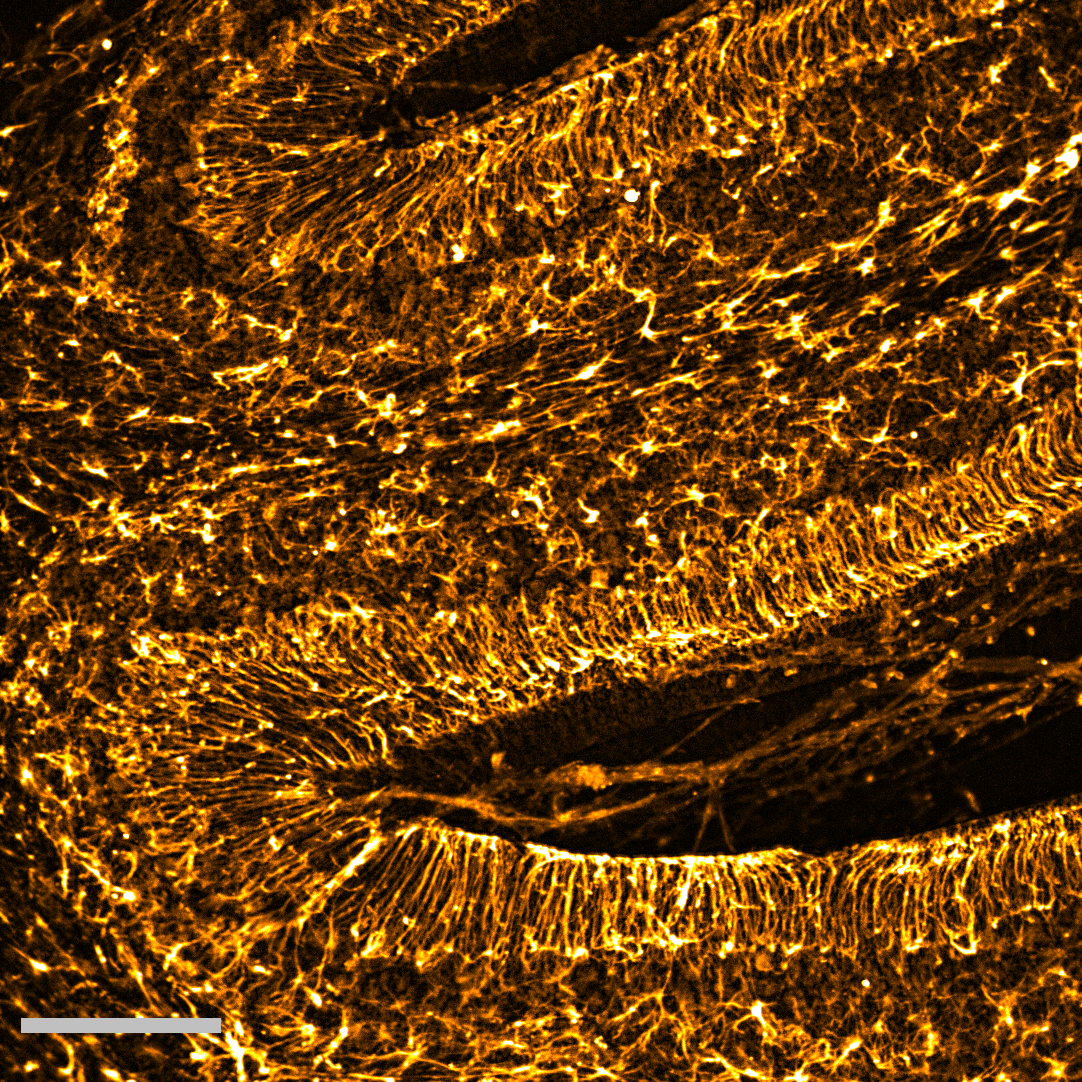
Being often overlooked, glial cells play multiple key roles in the brain: from governing nutrient supply to the brain to controlling the electrical signalling of neurons. In this image a type of glial cells called astrocytes, a.k.a the resident stars of the brain (from the from Ancient Greek ἄστρον, ástron, “star“) are shown in golden. These astrocytes are labelled in an acute section of a mouse cerebellum – a region known as “the little brain” which regulates movement and coordination. Importantly, recent studies show that specialised astrocytes in the cerebellum might be the reason why this brain region is resilient to multiple neurodegenerative conditions. |
Committee Vote: Through Cancer's Core
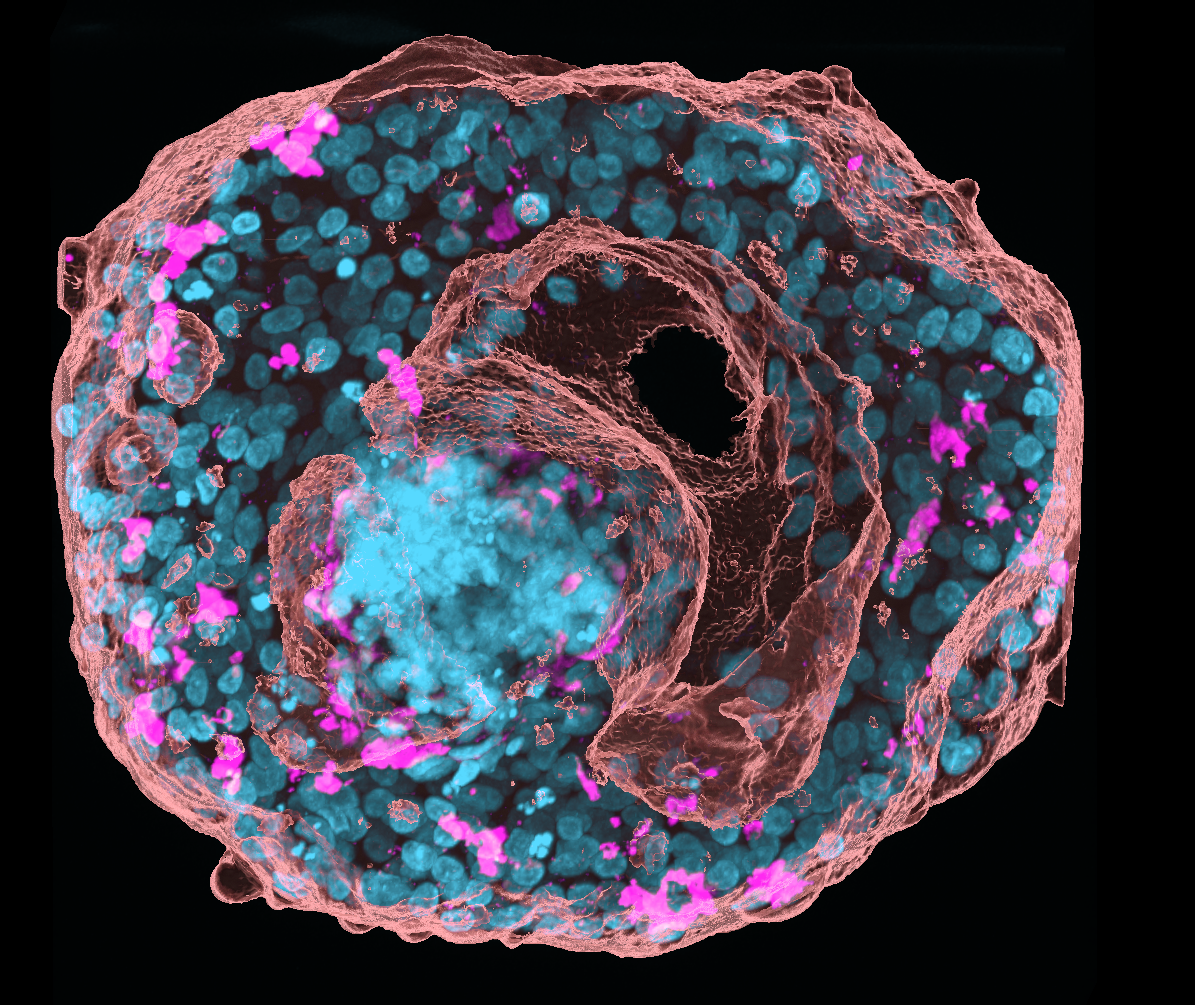
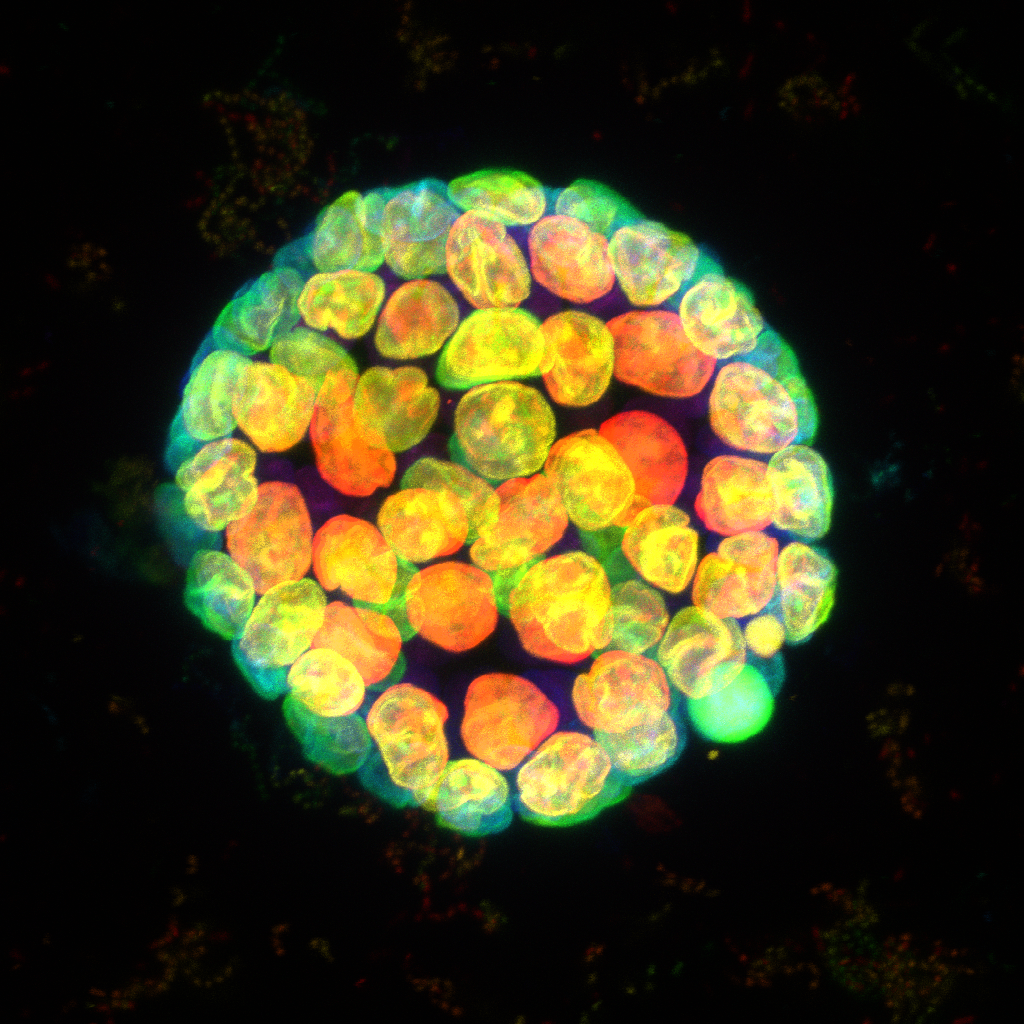
Pancreatic adenocarcinoma (PDAC) is an aggressive cancer that often metastasises to areas such as the liver. To study this metastasis we are developing a 3D in vitro model using a co-culture system. This image shows a spheroid generated by the aggregation and culture of multiple liver cell types and PDAC cells. To study potential modelling of the extracellular matrix, the secretion of collagen 1 within the spheroid was visualised by immunocytochemistry. The spheroid was also stained with DAPI and Alex Fluor 568 Phalloidin, and a z stack image was taken using a Nikon A1R confocal microscope.
Other Competition Entries
1. Microvesicles in Motion
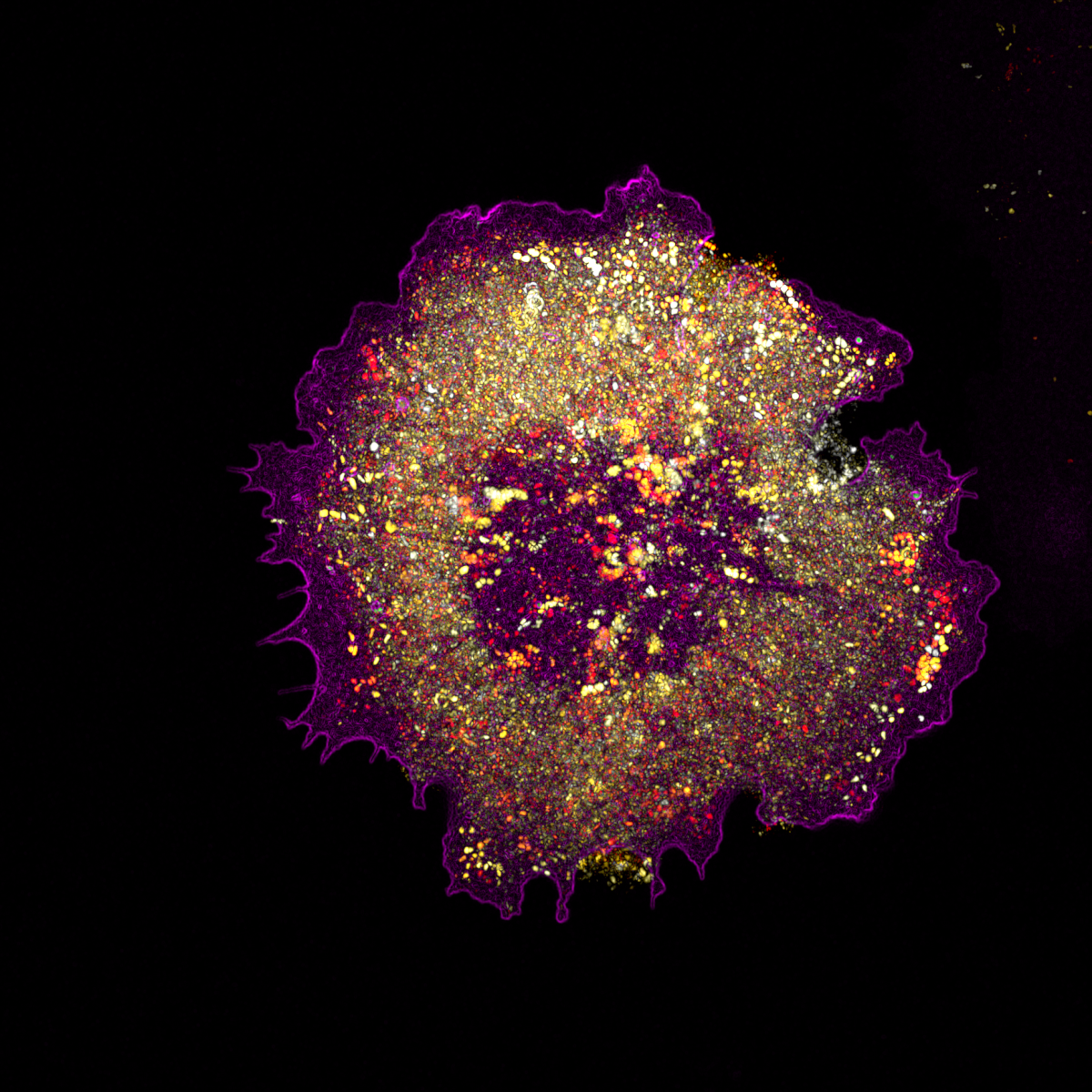
The image shows a cell expressing a marker for caveolae, a small endocytic pathway responsible for trafficking receptors and sensing the mechanical properties of the surrounding environment. The vesicles are represented as temporal colour code to show how dynamic they are, and all are enclosed within a cell (magenta). The image is taken using a super-resolution microscope where individual vesicle (dots) can be seen. |
2. Nebula
Taken using a Zeiss LSM710 confocal with a x20/0.8 objective. The sample is multispectral beads and was taken as part of a CLEM project using MatTek glass bottomed gridded dishes. I was testing a new x5 phase objective to give an overview of the grid (as this was not visible with DIC). I was using the beads as fiducials to correlate between fluorescence and the Electron Microscope. But came across a patch of beads that had not dispersed properly and became side tracked obtaining this image.
6. Spectral Flower
This image is the result of a test of highly multiplexed spectral imaging. Small drops of mounting medium containing 9 different fluorescent markers were placed on a glass slide and imaged together using a confocal Zeiss LSM 880 microscope with spectral detection. Signal was then unmixed to create a channel for each dye. Autofluorescence from air bubbles (gray) was unmixed as an independant channel. |
7. The Complex Landscape of the Mouse Salivary Gland
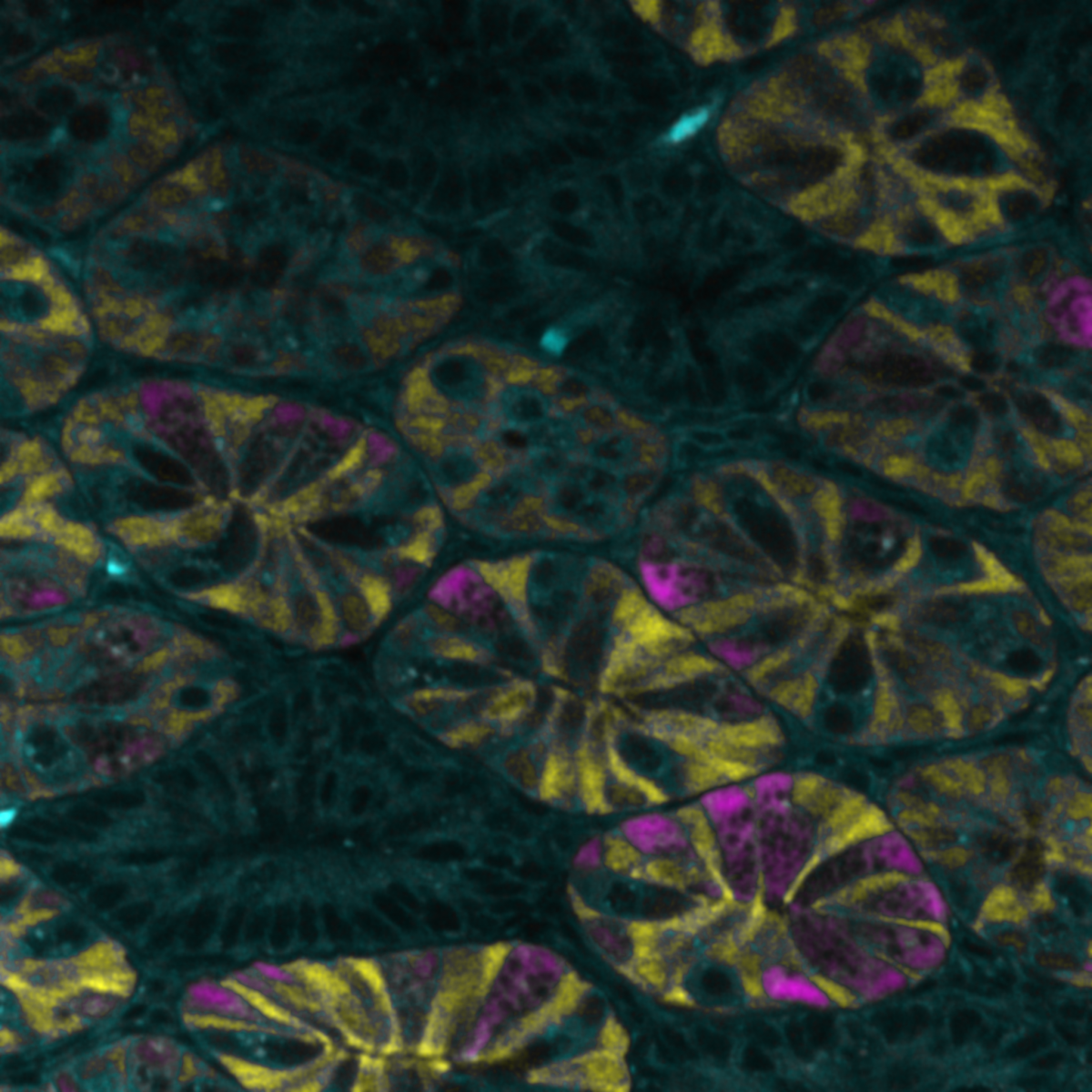
The image shows a section of a mouse submandibular salivary gland imaged on a Leica SP8 confocal microscope (The Centre for Regenerative Medicine, The University of Edinburgh). The magenta shows the ducts (stained with the epithelial marker E-cadherin) which transport the saliva, while the cyan colour shows the saliva producing acini (stained for the water channel Aquaporin-5), which make the saliva.
The salivary glands are inadvertently damaged during radiotherapy for head and neck cancer, leading to problems with speech, eating and to oral health issues. In order to develop regenerative strategies we must first understand the composition of the salivary gland and how different cell types respond to irradiation injury. Confocal microscopy is pivotal in characterising irradiation injury in both mouse and human salivary gland.
10. The Infected Cell Lounge
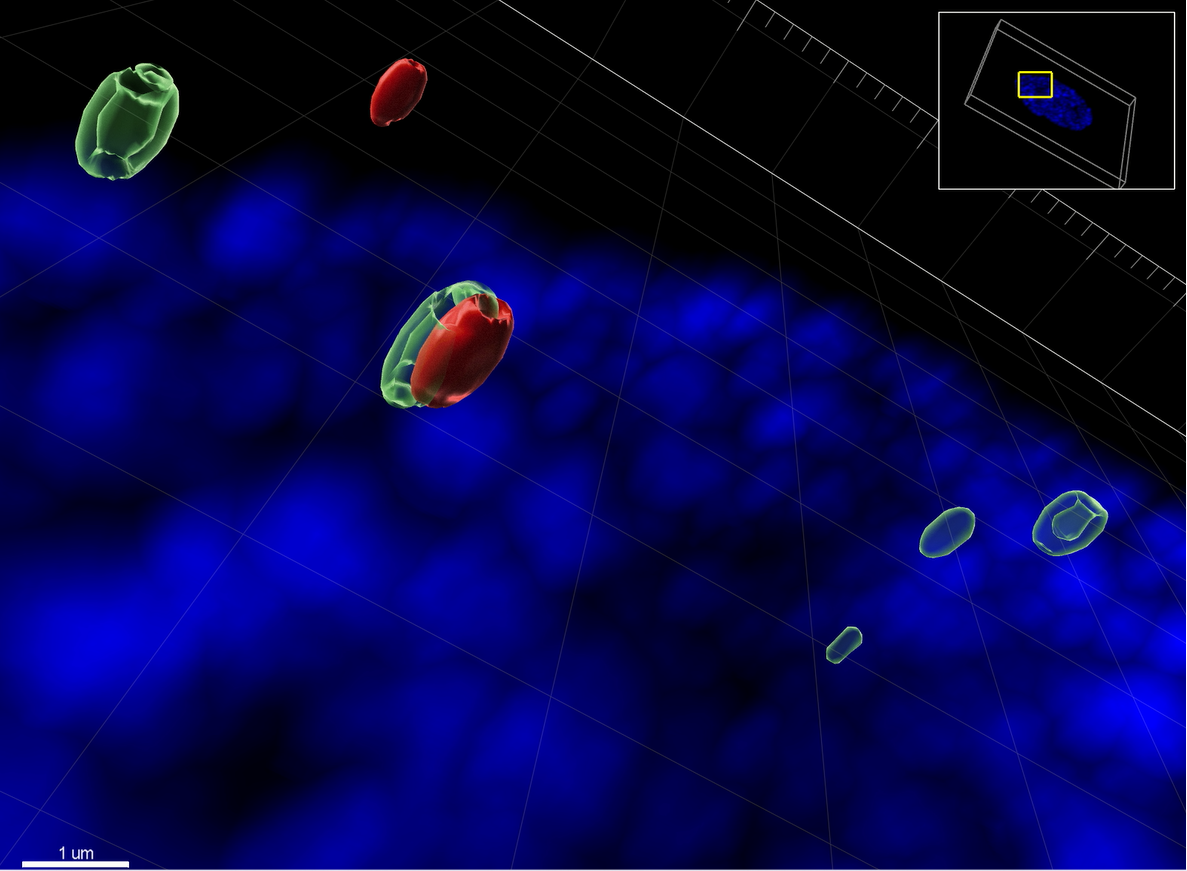
The confocal microscope ZEISS LSM 880 with Airyscan was used to image a Z-stack of a primary immortalized human fibroblast (HFt) cell, KO for PML, with the nucleus stained in blue with DAPI. The high resolution image was processed with IMARIS software to render the surfaces of two click chemistry labelled HSV-1 genomes in red, and the host cell protein Speckled-100 (Sp100) stained by immunofluorescence in green. Sp100 has nuclear speckled distribution and can associate with viral DNA genomes once they invade the nucleus. The labelled virus requires to be produced in advance by pulsing retinal pigmented epithelial (RPE) cells with modified nucleotides (EdC/A) that the virions incorporate over a period of 3 days. Then, the supernatant is collected, cleared by low speed centrifugation, filtered to remove cell debris, column purified to eliminate the excess of EdC/A, and the viral titre is determined. HFt are stably immortalized by expressing telomerase under hygromycin selection, and knock out (KO) for the major promyelocytic-leukaemia nuclear bodies’ (PML-NB) protein PML under puromycin selection, using a lentivirus system. It is known that PML can entrap viral DNA at 90 minutes after infection to repress its expression. PML KO HFt were infected with labelled HSV-1, fixed 90 minutes later, clicked and stained to shown that Sp100 hollow nuclear structures can also associate with viral DNA. Sp100 has several domains and motives to bind chromatin, or predicted to be involved in DNA binding. HSV-1 access the cell nucleus “naked”, and the cell use immunity factors as Sp100 or PML to assemble chromatin into it, attempting to keep the invasion expression repressed.
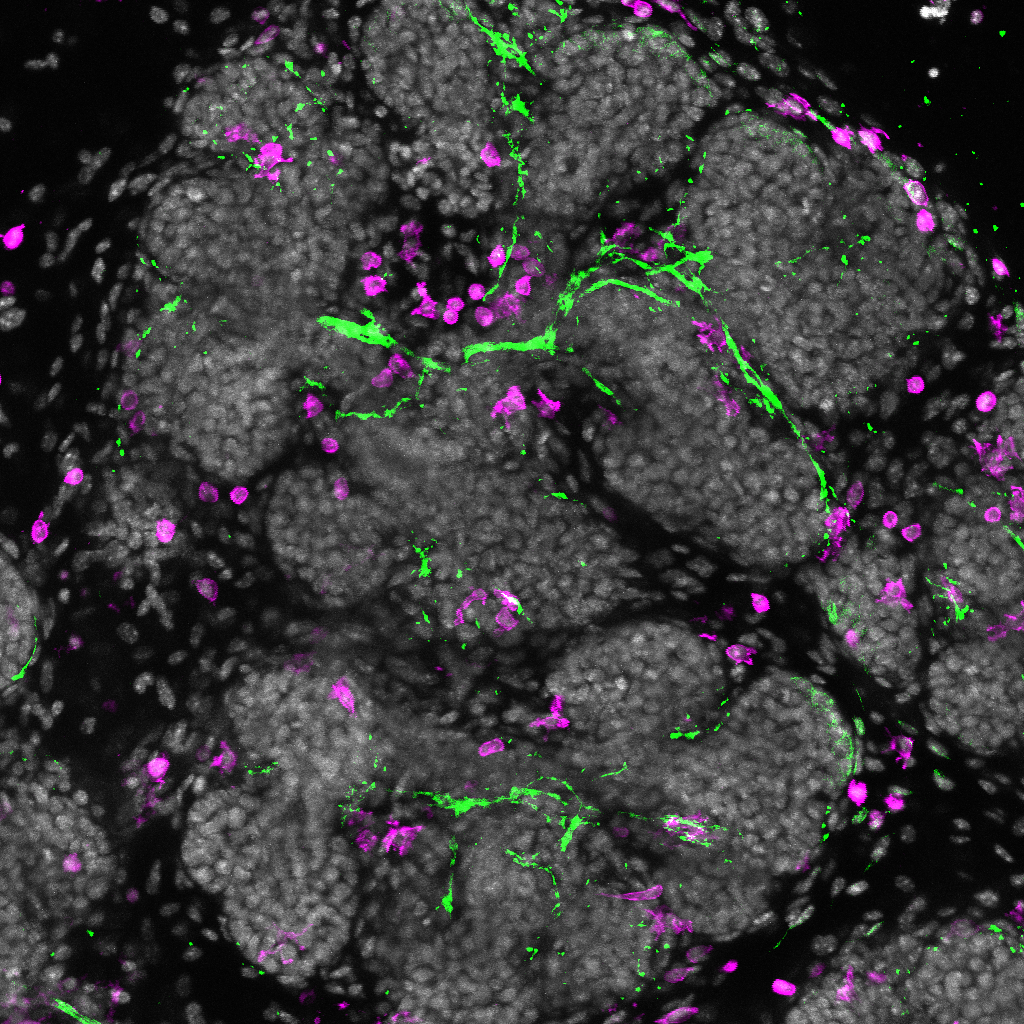
11. The Salivary Tree of Life
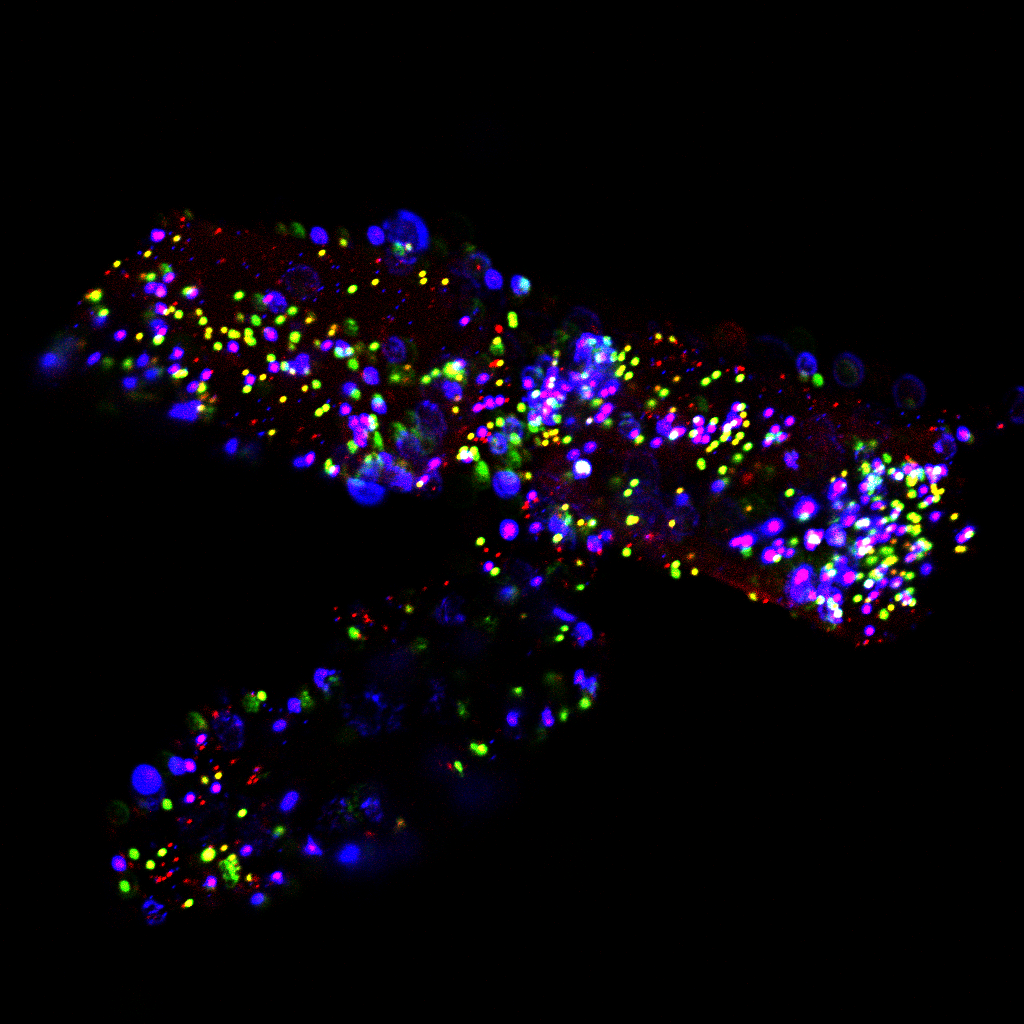
Max projection of wholemount embryonic (E15.5) mouse salivary gland immunofluorescently stained for nerves (green) and macrophages (magenta), with cell nuclei in gray. Imaged on a Leica SP8 Confocal microscope (Centre for Regenerative Medicine imaging core facility). |
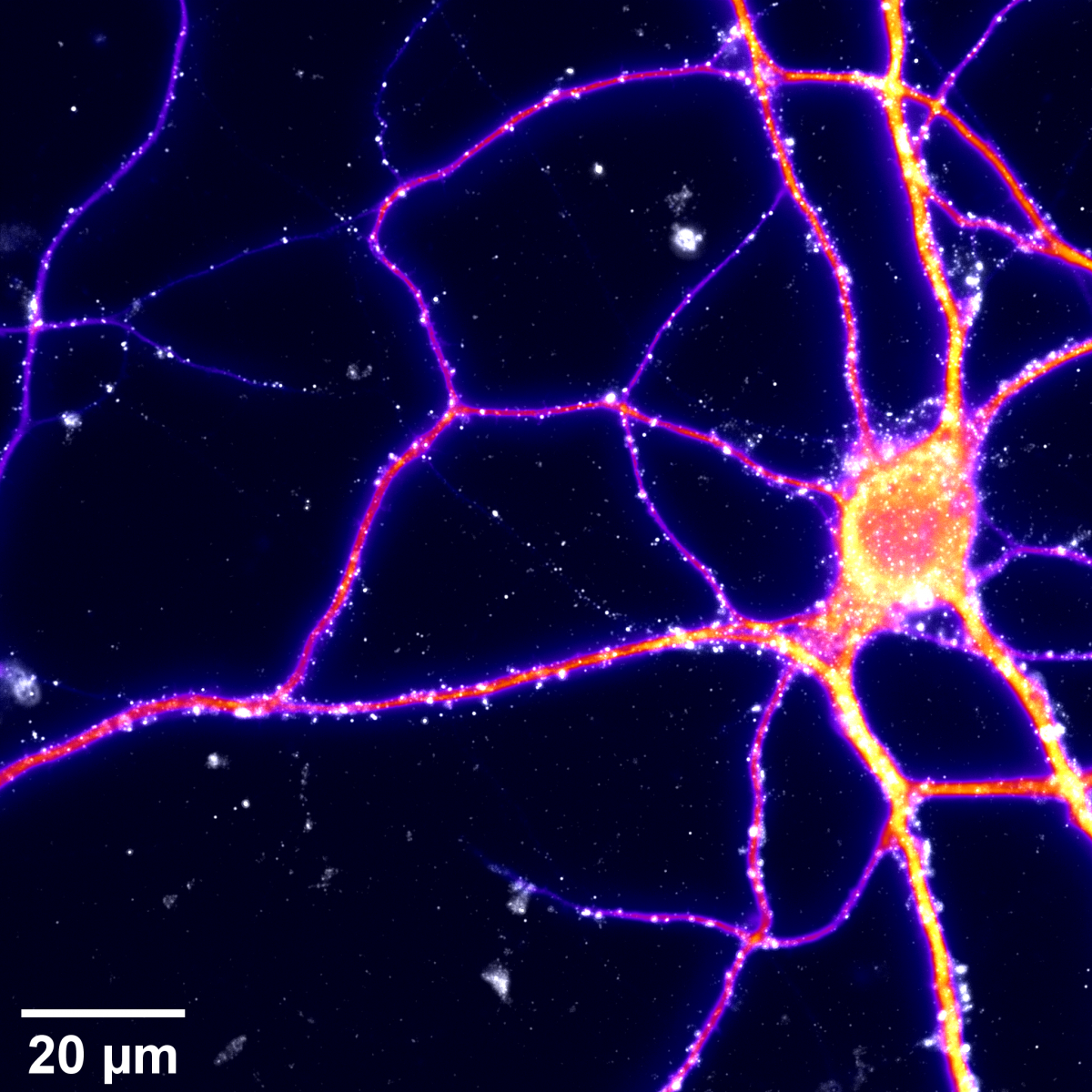
12. There is Another Galaxy
Mouse hippocampal neurons cultured at very low density using the Banker technique (using a glial mat feeding layer).
They were fixed at DIV12 and immunostained for MAP2 (Microtubule-associated protein 2 ; fire) showing primary and secondary dendrites, and PSD95 (Post-Synaptic Density protein 95 ; white-grey) expressed in dendritic spines and commonly used to label (post-) synapses.
Images were acquired in epifluorescence mode using an inverted microscope, exciting AF405 (MAP2) and AF532 (PSD95) fluorophores with a classic LED lamp.
Get in touch with the Scottish Microscopy Society!
Feel free to contact us or join our mailing list using the contact form below.